INano™ S
INanoTM S is a GMP-compliant commercial-scale production equipment that integrates upstream and downstream processes, including LNP encapsulation and TFF.
Overview
INanoTM S is a GMP-compliant commercial-scale production equipment that
integrates upstream and downstream processes, including LNP encapsulation and
TFF. This instrument has been successfully utilized for commercial-scale
production of COVID vaccines, demonstrating its reliability and efficiency in
meeting high-demand manufacturing requirements.
Flowrate range: 1-4 L/min (without dilution)
Designed with the QbD (Quality by Design) concept, allowing full
adjustability of key process parameters
Compatible with public utilities such as water-for-injection and
high-temperature steam for CIP/SIP, with a robust design to minimize
contamination risks
Offers flexibility to use microfluidic mixing chips or T-mix mixers without
the need for additional consumables.
Proven success in multiple commercial-scale production for LNPs.
Capabilities
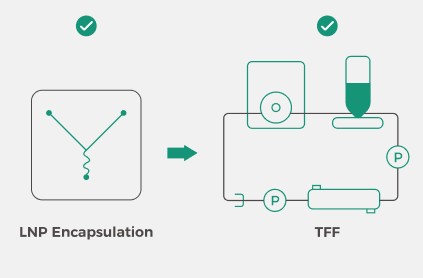
Integration of LNP Encapsulation and Downstream Processing
The Inano™ S is large-scale commercial GMP production equipment. It
innovatively integrates LNP encapsulation with downstream TFF purification and
concentration processes, achieving full process continuity and automation.
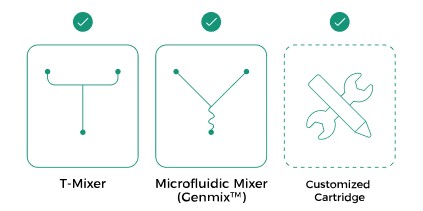
Multiple Mixing Methods
As a platform-based device, the INano™ S not only supports microfluidic mixer
and T-mixer structures, but also allows for the customization of mixing
structures based on customer requirements. Additionally, it offers a variety of
mixer materials for users to choose from, such as 316L stainless steel,
Hastelloy, and more.
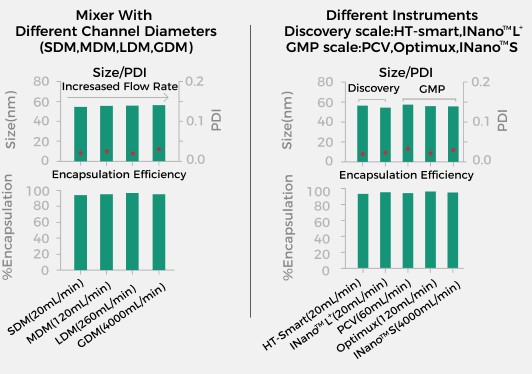
Consistent Critical Quality Attributes (CQA) Can Be Achieved From Preclinical
to Clinical Development
Ensure that all preparation process parameters remain unchanged to avoid
secondary process development, guaranteeing a high success rate for scale-up,
allowing you to easily become a process expert!
GMP Complicance
The equipment is meticulously designed and manufactured to comply with GMP,
cGMP, and EudraLex regulations. Adherence to ASME and BPE standards ensures
precision and quality throughout the process. Constructed with premium materials
and components, the equipment delivers exceptional stability and reliability,
meeting the highest industry standards.

Comprehensive Documentation and Full Traceability
The equipment's operating system is designed in strict compliance with FDA 21
CFR Part 11, incorporating multi-level user access management, data recording,
audit trails, electronic signatures, and may more additional features.

Related products
-
SCIENTZ08-IIIA Non-contact Ultrasonic DNA Shearing Instrument
Contact UsThe SCIENTZ08 – IIIA non – contact ultrasonic DNA shearing instrument uses isothermal, non – contact means to shear, homogenize and mix samples.
-
ELISA Plate Reader with CE and ISO Citified
Contact UsBig touch screen Elisa Microplate reader Suit for all kinds of ELISA reagents
-
TOC-5000 RN/RD Total Organic Carbon Analyzer for Water Analysis
Contact UsAuto sample dilution, auto acid-adding and auto gas purging (only TOC-5000 RD).METASH unique NDIR detector, German made light source, higher sensitivity and stability, longer service time.
-
Traffic Speed Radar Long Range Detection LDTR20
Contact Us● Frequency:24GHz
● Detection distance:≥200 meters
● Communication interface:supports RS485 and TTL communication methods
● Product form:integral radar with housing or separate PCBA (radar sensor)
● Customization service:Support personalized product customization






Reviews
There are no reviews yet.